Review
Modification of Carbon Nanotubes as an Effective Solution for Cancer Therapy
Sara Tavakolifard 1, Esmaeil Biazar 2*
1 Department of Chemistry, Science and Research, Yazd University, Iran.
2 Department of Biomaterials Engineering, Tonekabon Branch, Islamic Azad University, Tonekabon, Iran.
* Corresponding author. E-mail: kia_esm@yahoo.com
Received: Jul. 13, 2016; Accepted: Sep. 7, 2016; Published: Sep. 15, 2016.
Citation: Sara Tavakolifard, Esmaeil Biazar, Modification of Carbon Nanotubes as an Effective Solution for Cancer Therapy. Nano Biomed. Eng., 2016, 8(3): 144-160.
DOI: 10.5101/nbe.v8i3.p144-160.
Abstract
Carbon nanotubes (CNT) as a new class of nano-materials hold great potential for various biomedical applications. Owing to their unusual properties, carbon nanotubes have been extensively employed in electronics, nanotechnology and optics, among others. In spite of the great potential of carbon nanotubes in various domains of biomedicine, inefficient dispersion in aqueous solutions and biological activities in vivo are still disputable. One important and feasible route in a struggle to overcome these obstacles is modification of CNTs with organic compounds and polymers, which have been widely studied and play a crucial role in biological and biomedical fields, particularly in the cancer therapy. This review focuses on the breakthrough of the recently used methods to functionalize onto the surface of carbon nanotubes with multiple chemical species in order to produce anticancer drug delivery systems for biomedical applications.
Keywords: Drug delivery; Cancer therapy; Carbon nanotubes; Biological transporters; Targeting
Introduction
It has been observed that advances in functionalization chemistry have been considered as a driving force in the development of new classes of new nano-materials for application in biology and medicine in the past decade [1–24]. The employment of Carbon nanotubes (CNTs), with their unique physical and chemical properties, is promising in the development of new generations of anticancer drug delivery systems. Modifications of CNTs by polymers through covalent and non-covalent bonding is one of the best approaches to adapt the collaborations of the Carbon nanotubes with environmental components together with solvent and cells. Notably, CNTs have attracted considerable interests in this regard, as they offer the potential collaborations between CNTs and polymers which are important as newly made hybrid nano-materials being simply able to cross cell membranes without any adverse biological response and, furthermore they are able to deliver a wide range of biologically active molecules containing drugs and genes into the cells. CNTs offer potential advantages over the more widely studied various nanoparticle systems, their ability to carry a high cargo loading, their intrinsic stability and structural flexibility, which could prolong the circulation time and hence the bioavailability of the carried drug molecules [25-28]. Drug delivery is an emerging field focusing on a wide range of biologically active molecules including drugs and genes for a desirable group of cells. The goal of this targeted delivery is to transport a proper amount of drugs to the desirable sites such as diseased tissues, with minimizing unwanted side effects of the drug on other tissues. The development of targeted therapy represents an exciting and a new approach to cancer treatment, and due to their promising properties the targeted drug as a responsive molecule attached to specific surface receptor proteins located in cell membranes [29-31]. Being endowed with targeting specificity, the carrier is able to focus just on the looked-for biological area. The use of these biological carriers for the development of specific and sensitive site-targeted bio-nano-systems makes the selective internalization of molecules in cancer cells possible. This internalization process is not feasible under normal conditions [32,33]. Hence, such bio conjugation holds remarkable potential for future cancer treatment. Bio conjugation makes a base for further studies on cancer treatment in the future. Despite excellent progress in using CNTs as drug delivery vehicles in bio functionalization of CNTs using various biological molecules such as polyethylene glycol (PEG), DNA, antibodies, folate, cytostatic drugs, and growth factors [34-37], there is a significant lack of knowledge on how to obtain selectivity of these compounds for a single type of cancer cell. This shortage is due to the simultaneous presence of the receptors used for the specific binding of the targeting molecules to the membranes of the noncancerous cells, even in smaller amount than the cancer cells [38-40]. In this review, we focus on summerizing the latest progress in the use of CNTs for covalent and non-covalent interactions with various types of biological species, as well as bio macromolecules interactions which are very crucial to descending the toxicity of CNTs in order to study the in vitro selective drug delivery of cancer cells.
Physical and Chemical Properties of the Carbon Nanotube
More allotropes of carbon have been reported and a large scientific community has been passionate about deciphering the properties of this element that can adopt many structures ranging from diamond and graphite (three-dimensional) to graphene (two-dimensional), nanotubes (one-dimensional) or fullerenes (zero-dimensional). In this way, CNT is analogous to fullerenes and graphene, which vary in wall number, diameter, length and surface chemistry [41]. Carbon nanotubes were first discovered in 1991 by Sumio Iijima in Japan [41]. Carbon nanotubes are well-ordered, hollow nanostructures consisting of a hexagonal arrangement of sp2 hybridized carbon atoms (C-C distance is about 1.4 Ǻ) which are stronger than sp and sp3 bonds and are the key factors rendering CNTs excellent mechanical strength and high electrical and thermal conductivity. Conceptually, Graphene is the two-dimensional, hexagonal sp2-carbon ring networks with one atomic layer thickness, while CNTs can be imagined to form when the graphene sheet is rolled to be held together by van der Waals into a cylinder [42, 43]. One rolled-up graphene sheet will form a single walled carbon nanotube (SWCNT), while more than one concentric graphene sheet create a multi-walled carbon nanotube (MWCNT). Single wall carbon nanotubes have diameters ranging typically from 0.5 to 1.5 nm and a length ranging from 100 nm up to several micrometers. MWCNTs have larger diameters (can be more than 100 nm) due to multilayer structures. The interlayer separation of the graphene layers of MWCNTs have a larger outer diameter (2.5 to 100 nm) than SWCNTs (0.6 to 2.4 nm); additionally, SWCNTs have a better defined wall, whereas MWCNTs are more likely to have structural defects, resulting in a less stable nanostructure. Among the various types of nano-materials (that is, materials that have at least one dimension less than 100 nm), there has been intense interest in the allotropes of carbon due to their unique physical and chemical properties, emerging as promising candidates for a multimodal drug delivery systems and early diagnosis of cancer cells [42, 43]. Owing to their large surface area, CNTs may be conjugated with a wide variety of ligands, such as proteins [44], peptides [45], enzymes [46], polysaccharides [47] and drugs [48]. These molecules can be bound to the CNT covalently or non-covalently (Figure 1) [50]. These nano materials are hydrophobic in nature and thus insoluble in water, which muzzles their application in biomedical and medicinal chemistry [48]. Depending on the type of conjugated polymer, modified CNTs can be soluble in aqueous or organic solvents. In order to avoid the aggregation of CNTs, to render their hydrophilic characteristics and to facilitate their use for applications in drug delivery, biosensors, tissue engineering, cancer imaging, and thermal, various functionalization methods like adsorption, electrostatic interaction and covalent bonding are utilized with a number of compounds and polymers [49,50]. Compared with CNTs, graphene exhibits some important qualities such as low cost, facile fabrication and modification, the higher surface of area with two external surfaces and the absence of toxic metal particles which threaten the dominance of CNTs in many applications, including drug delivery, demonstrating lower toxicity and superior biocompatibility [51].
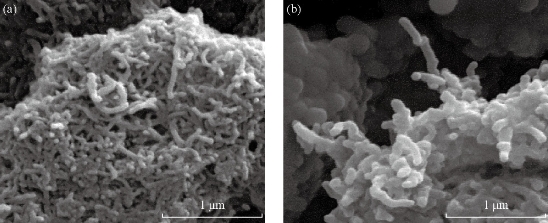
Fig.1 (a) Scanning electron microscope images of SWCNT; (b) and modified SWCNTs with Taxol/Folic acid [50].
Applications of CNTs in Cancer Therapy
Currently, available technologies have made enormous advancement in cancer research, but an adequate therapy remains elusive [52]. Cancer is characterized as a disorder which arises following a number of mutagenesis steps, allowing cancerous cells to grow and divide uncontrollably. These processes allow cancer cells to acquire properties of unlimited proliferation potential, self-sufficiency in growth signals and resistance to both anti-proliferative and apoptotic cues which would, otherwise, contain their growth. Tumors have also evolved methods to gain further support through interactions with surrounding stromal cells, promoting their angiogenesis, their evasion of immune detection and their metastasis to distal organs [52, 53]. The Global Cancer Report issued by the World Health Organization (WHO) estimates that there are over 10 million new cases of cancer every year with over 6 million annual deaths caused by the disease [54]. The most frequent types of cancers causing death are, as listed by the WHO: lung, stomach, liver, colon and breast cancers [54]. Current cancer therapies can be broadly categorized into two groups: cytotoxic therapies and molecular targeted drugs. Examples of traditional cytotoxic therapies include radiation and chemotherapeutic compounds such as platinum-based drugs [55-57]. Radiation, utilizing photons, damages the DNA of cancer cells by creating free radicals, leading to the inhibition of cell division and, eventually, causing cell death. The current medical anticancer treatments involve applications where therapies are provided by removing the diseased cells (surgery), burning them out (radiation therapy), or poisoning the diseased cells faster than the healthy cells (chemotherapy), most of which do cause damage to healthy cells [58-61]; moreover, present treatment methods are not very effective at stopping the spread or recurrence of cancer. One approach to overcome these drawbacks is design of nano-carriers which actively able to bind to specific cells after extravasations. This binding may be achieved by attaching targeting agents to the surface of the nano-carrier by a variety of conjugation methods. Then, nano-carriers will be recognized and bind in order to target cells through ligand–receptor interactions [62,63]. A wide variety of ligands can potentially be employed to target cellular biomarkers including antibodies, [47] other proteins [47], peptides [45] and polysaccharides [46]. Improved targeting of carbon nanotubes (CNTs) to tumors is essential because larger tumors exhibit poor vascularization, particularly inside necrotic areas, resisting the localization of CNTs in the tumor. Additionally, no solvent is required for delivery of the drug, which means that undesired health effects from the solvent can be prevented; as well as, a lower concentration of the toxin is delivered to other parts of the body. Hence, the aftermath of chemotherapy descend striking by the patient undergoing treatment. Carbon nanotubes can also be used for hyperthermia treatment of cancer. The innate optical properties of CNTs, such as strong resonance Raman scattering and near infrared photoluminescence (NIR PL) in the 1.1–1.4 µm spectral region, make them useful biological imaging agents [64-68]. To image the nanoparticle uptake into tumors, through the enhanced permeability and retention effect (EPR) [69] is a key to nanomaterial-based cancer nanoparticles, such as nanotubes and gold nanoparticles, have been used as NIR contrast agents [70,71]. Cancer cells are more susceptible to heat increases than healthy cells owing to the fact that the cancer cells are surrounded by blood vessels that have a strange flow result in dissipating heat less efficiently than blood around healthy cells [72]. However, to minimize the damage to healthy cells, heat still needs to be selectively delivered to tumor cells. The near infrared (NIR) (700 to 1100 nm) is a special region where optical absorption in tissues is minimal and penetration is optimal and carbon nanotubes have a strong absorption in this region [73,74]. Therefore using NIR light can effectively heat up CNTs in vivo. The high aspect ratios of CNTs are suitable for attaching some ligands such as folic acid with the aim to recognize folate receptors overexpressed on many tumors, including ovarian, breast, brain, kidney, lung, and liver [47]. Another multicomponent CNT-based system for cellular imaging applications in cancer therapy and magnetically guided drug delivery for local hyperthermia illustrate great undertaking in the harmless and effective transducer for cancer treatment. Magnetic nano-materials (MNP) based hyperthermia treatment has a number of merits compared to conventional hyperthermia treatment. In this method cancer cells absorb MNPs, thereby increasing the effectiveness of hyperthermia by delivering therapeutic heat directly to MNPs, can be targeted through cancer-specific binding agents, as a result of which it makes the treatment much more selective and effective. Thus, the rate of drug releasing can be controlled by directly modulating the magnetic field. In a nutshell, a number of types of magnetic nano-materials, ranging from well-known and well-investigated iron oxide-based nano-materials to metallic NPs such as Mn, Fe, Co, Ni, Zn, Gd, Mg, and their oxides have been investigated for their hyperthermic potential. Some of the hyperthermic agents based on iron oxide are magnetite NPs (Fe3O4) stabilized by a variety of ligands such as dextran, [75-77] polyvinyl alcohol, hydro-gel and magnetic NPs (γ-Fe2O3) [78,79], cationic liposomes, [80] stabilized with hydrophilic polymers such as starch or dextran and chitosan [81,82]. Since the targeted therapy has the characteristics of driven magnetic accuracy, targeting, and high drug-capacity, it can be effective to lower toxic effects and to enhance the therapeutic effect [43, 44]. Although, all these properties make this an ideal method for drug delivery, selectively targeted drug delivery systems are still facing challenges including improvement of specificity, stability, regulating bioavailability, and developing targeted carriers with lower toxicity [47-65].
Internalization of CNTs, Cellular Uptake of CNTs
Many studies have confirmed the cellular absorption of CNTs. Nonetheless, it is however, not clear how CNT penetrates into cells. Successful chemotherapy, therapeutic agents should pass a series of biological barriers including hepatic and renal clearance, enzymolysis and hydrolysis, as well as cellular uptake and endosomal/lysosomal degradation [83-85]. Various mechanisms for the internalization pathway of SWCNTs inside living cells have been proposed. Pantarotto et al. [84] suggested that insertion and diffusion of nanotubes across cell membranes is an energy-independent non-endocytotic process. In contrast, other reports have indicated that cell membrane transport of SWCNTs conjugates with proteins and DNA is through the Clathrin-receptor (mediated) dependent endocytosis pathway [86]. In a study conducted by Kam et al. [87], protein attached to SWCNTs-biotin was detected in the endosomes, suggesting that nanotubes uptaking occurred via endocytosis. It has been shown that MWCNTs and single-walled carbon nanotubes (SWCNTs) differ in their cell penetration mechanism. Additionally, the size of the CNTs influences their cellular uptake and fatality of cells, owing to the fact that long SWCNTs are localized in the cytoplasm, whereas short SWCNTs are transported into the nucleus, whereas the MWCNTs are excluded from the interior (Figure 2) [88]. It has been proved that a proficient method is needed to minimize the noxious effects and also to increase the level of therapeutic response to CNTs as represented by their conjugation to a carrier molecule [89,90]. For instance, various strategies for the fabrication of nano moieties to target folate receptors on the cancer cell membrane have been proposed [90]. Folate receptors are overexpressed in cancer and their targeting allows CNTs to facilitate cellular internalization of folate containing species by receptor-mediated endocytosis which is more selective than CNTs alone that enter cells through phagocytosis or endocytosis and through passive diffusion [91]. Most researchers have focused on the potential of CNTs for delivery of anticancer agents which might be attributed to their exclusive physicochemical features especially their ability for crossing various biological barriers within the body without generating an immunogenic response and toxic effects [92].
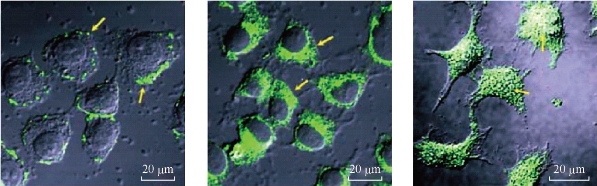
Fig.2. Confocal microscopy images show that the ability of MWCNT, S-SWCNT and L-SWCNTs to be internalized into the cells, whereas the MWCNTs are excluded from the interior [88].
Functionalization of CNTs
Pristine, CNTs’ inner and outer surfaces have different characteristics, offering the possibility of loading the inner space with biologically active species; moreover, modifying the outer surface to make the biocompatible nanotubes They tend to bundle up and are insoluble in most types of solvents [93] making it difficult to use them in biomedical and medicinal chemistry. In addition, some CNTs without any functionalization have been shown to be cytotoxic [94-96]. Furthermore, bioactive agents can be conjugated to CNTs through functionalization, which as a result can serve as a drug carrier for drugs, antigens and gene delivery. Modification reactions of CNTs can be classified into two main approaches: (i) oxidation followed by carboxyl based couplings (ii) additional reactions to the sidewalls and tips of CNTs or non-covalent reactions. In the first and most important approach, functionalization strategy consists of oxidation and carboxyl-based coupling. The carboxylic groups also allow covalent couplings with other molecules through amidation, esterification, thiolation, halogenation, hydrogenation, and cycloaddition reactions as well as radical, carbene and electrophilic additions [44]. The second strategy is through additional reactions employed to attach some organic groups to the sidewalls and/or tips of the CNTs. Strategies for non-covalent modification include the use of: (a) organic solvents [97], (b) organic polymers [98,99], (c) amphiphilic peptides, (d) nucleic acids [100], sugars, and (e) detergents [101]. One of the largest applications for drug delivery is in cancer treatment, where proper amounts of drugs need to be directed to the targeted tissue while minimizing unwanted effects of the drugs on normal tissues. Several water-soluble polymers have been covalently and non-covalently attached onto the surface of CNTs to produce modified water-soluble CNT–polymer (Table 1). Depending on the type of conjugated polymer, modified CNTs can be soluble in aqueous or organic solvents, for example, conjugation of polylactide or polycaprolactone onto the surface of CNTs tends to solubilization of CNTs in organic solvents such as chloroform [102,103]. It has been proved that polymers not only raise the functionality, biocompatibility and water solubility of CNT but also are able to change the CNTs conformations dramatically [102-104]. According to CNTs’ polymeric and cationic innate abilities, soluble functionalized carbon nanotubes seem to represent an innovative drug delivery system, since they are capable to penetrate into the cell without altering their morphology [105,106]. It is well worth noting that functionalized carbon nanotubes with Fluorescein isothiocyanate (FITC) or a fluorescent bioactive peptide peptide are able to cross the cell membrane, while the CNT with the FITC mainly distributed into the cytoplasm and slowly moved into the nucleus, the CNT with the fluorescent peptide rapidly penetrated into the nucleus. These CNT conjugates entered the cell with a passive mechanism. In fact, the translocation capacity was not modified by decreasing the temperature or by using inhibitors of endocytosis-mediated process. One strategy to specifically deliver drugs to a tumor site is through the enhanced permeability and EPR [34]. The EPR effect is due to leaky vascular structures and an impaired drainage system of tumors. Therefore, nanoparticles with sizes ranging from 100 to 700 nm cannot penetrate the pores of the blood vessels in healthy tissue but will go through the pores of tumor blood vessels and preferably accumulate at the tumor site [107,108]. CNTs can be conjugated with a therapeutic agent and will preferably accumulate at the tumor through the retention effect [109-118].
Table 1. Carbon nanotube based targeted delivery system
|
Ref. |
Tumor |
Targeting agent |
Drug |
Compound |
|
50 |
Breast cancer; Cervical cancer |
FA, FITC |
PTX |
SWCNT-PTX-FA-FITC |
|
108 |
_ |
FITC |
MTX |
MWCNT-MTX |
|
109 |
Breast cancer |
_ |
PTX |
SWCNT-PTX-PEG |
|
110 |
Breast cancer |
RGD |
DOX |
SWCNT-PL-DOX |
|
111 |
Liver cancer |
FA |
DOX |
SWCNT-CHI-DOX |
|
112 |
Cervical cancer |
FA |
DOX |
SWCNT-ALG-CHI-DOX |
|
113 |
Breast cancer |
FA |
DOX |
SWCNT-PEG-DOX |
|
114 |
_ |
FA, QD |
PTX |
MWCNT-PEG-PTX |
|
55 |
_ |
FA |
cisplatine |
SWCNT-PEG-Pt (IV) |
|
115 |
Colon adenocarcinoma |
_ |
cisplatine |
MWCNT-PCA-PEG-PCA |
|
116 |
_ |
_ |
PTX |
MWCNT-PCA-PTX |
|
117 |
Leukemia |
FITC |
Taxoid |
SWCNT-FITC-Biotin |
|
118 |
Colon cancer |
FITC
|
DOX |
SWCNT-BSA-FITC-DOX |
Non-covalent method
Non-covalent attachments have received much interest owing to the possibility of linking different groups of polymers on the surface of CNTs without disturbing their bonding network [119]. One of the main problems in using CNTs as drug nano-carriers is their recognition as foreign particles in the bloodstream. Non-covalent adjustment approaches of CNTs have mainly been set up on the three general path ways comprising hydrophobic connections between the surface of CNTs and long-chain hydrophobic polymers, π-π interactions between CNTs and polymers containing a sector with the ability of producing ‘‘π-π stacking’’ interactions such as pyrene, ferrocene and other aromatic containing molecules [119,120], finally electrostatic and hydrogen-bond interactions between CNTs or acid-treated CNTs (-COOH) and macromolecules or polymers with polar functional groups which is named ‘‘polymer wrapping’’ mechanism [121]. Polyethylene glycol (PEG) is one of the most popular CNT surface modifiers, due to its hydrophilicity, flexibility and biocompatibility [122]. PEG-modified CNTs may also be dispersed in relatively higher salt-concentrated aqueous solutions [123]. The optimized CNT-PEG conjugate afforded relatively low reticuloendotheelial system (RES) accumulation, high tumor uptake and low skin retention, thus making CNT an ideal choice for in vivo cancer treatment [124]. The cellular internalization of the CNTs depends, among other factors, on the PEG-chain length; the longer PEG chains reduce cellular uptake [125]. In such an assay, large surface areas exist for supra-molecular chemistry on single-walled carbon nanotubes pre-functionalized non-covalently or covalently by common surfactant or acid-oxidation routes. Water-soluble CNTs with PEG-functionalization via these routes allow surprisingly high degrees of π-π stacking of aromatic molecules, including a cancer drug with ultrahigh loading capacity, a widely used fluorescence molecule (fluorescein), and combinations of molecules. Liu et al. [109] have shown that there is much space for supra-molecular chemistry assembly of molecules on SWCNTs pre functionalized non-covalently by common surfactant or acidoxidation routes. Aqueous soluble SWCNTs with PEG functionalization by these routes allow for π-π stacking of various aromatic molecules, including doxorubicin (DOX) as a cancer chemotherapy drug (DOX) with an ultrahigh loading capacity of _400% by weight and a widely used fluorescence molecule (fluorescein) [126]. Biologically inert SWCNTs with non-covalent PEG functionalization (PL-PEG5400NH) have been conjugated to antibodies such as Rituxan to selectively recognize CD20 cell surface receptor on B-cells with little nonspecific binding to negative T-cells and Herceptin to recognize HER2/neu positive breast cancer cells [109]. These results have suggested that unoccupied surface areas on functionalized SWCNTs were useful for binding of other molecules. It is suggested that non-covalent binding of DOX on SWCNTs most likely occurred via π-π stacking [127] and hydrophobic interactions owing to the aromatic nature of the DOX molecule and the relatively low solubility of deprotonated DOX at basic conditions. Free, unbound DOX exhibited high fluorescence, while weak fluorescence was observed for DOX after binding to SWCNTs. This high degree of fluorescence quenching is evidence of π-π stacked DOX, similar to other aromatic molecules π-π stacked onto nanotubes [126-128]. This represents a novel partition and utilization of the SWCNT sidewall surface area affording a unique and interesting way to attach drugs to carbon nanotubes in view of their extended polyaromatic sidewalls. In another investigation to demonstrate the targeted delivery of doxorubicin by SWCNTs, they conjugated a cyclic RGD peptide on the terminal groups of PEG on SWCNTs, imparting a recognition moiety for integrin receptors up-regulated in a wide range of solid tumors. RGD-labeled PL-PEG5000NH2 has been covalently used for loading of anticancer drug, doxorubicin, on the surface of SWCNTs to kill glioblastoma and breast cancer cells. The loading interactions between DOX molecules and SWCNTs surface is strong enough to prevent rapid desorption in normal physiological conditions. Molecular release of the non-covalently bound molecules can be triggered by environmental changes such as PH or other external stimuli. This system has also been used for loading/ releasing other aromatic molecules such as fluorescent dye molecules Fluorescein isothiocyanate and another aromatic chemotherapy agent (Doxorubicin) [129]. Another frequently-studied non-covalent interaction is a targeted drug delivery system based on polysaccharide [sodium alginate (ALG) and chitosan (CHI)] modified carboxylate SWCNTs for controlled release of DOX; in addition, including folic acid as a targeting agent [130]. The CHI/ALG-SWCNTs were initially oxidized and cut and then the appropriate polysaccharide (ALG or CHI), leading to the encapsulation of the SWCNTs by the polysaccharides [129- 132]. According to the polymer wrapping process involving initial treatment with ALG followed by CHI, the SWCNT core could be doubly wrapped by both CHI and ALG. Afterwards, CHI/ALG-SWCNTs with DOX in aqueous solution also allows the DOX to be attached to the nanotubes non-covalently [133]; moreover, chitosan reacts more efficiently with FA via an amidation reaction offering a further advantage over the polymer coated nanotubes in that one polymer has an affinity for the DOX drug and one for the FA targeting molecule. Consequently, the amount of DOX loading was monitored by ultraviolet-visible (UV-Vis) absorption spectroscopy [121]. According to the zeta potentials data of the modified SWCNT the positively charged DOX molecules are more readily absorbed onto the surfaces with lower surface potentials, suggesting that electrostatic interactions as well as π-π stacking interactions play an important role with respect to DOX loading [121]. Cell viability test results indicate that incubations with the unloaded nanotubes or the free drug result in appreciable cytotoxicity at the administered concentrations, indicating that the DOX-FA-CHI/ALG-SWCNTs are not only cytotoxic, but also selective. It is suggested that the DOX has been released in the low PH environment of the lysosomes, then migrating into nucleus to bind DNA. This latter interaction has been revealed to inhibit transcription and ultimately leads to cell death [133-135].
Covalent method
The use of carbon nanotube covalent functionalization to deliver anticancer agents has also been recently explored. The functionalization of carbon nanotubes for biomedical applications involves covalent modifications. Covalent modifications are based upon linking of already functionalized or commercially available polymer chains onto the surface of CNTs by suitable chemical reactions [136]. In biological applications, oxidation, amidation, esterification, radical coupling, and grafting polymers on the sidewalls of carbon nanotubes are widely adopted. However, in the grafting approach, first appropriate functional groups are created on the surface of CNTs, and then, desired polymers will be grown on the nanotube surface via polymerization reactions [136]. One of the recently reported example of this category is about effective pointing of an anticancer is conjugated methotrexate (MTX) by modified MWCNT. It has been suggested that the covalent functionalization of MWCNTs with MTX was achieved via a 1,3-dipolar cycloaddition approach [137]. In this survey, a methodology was developed for the introduction of two linking sites on the sidewalls of carbon nanotubes for FITC and MTX sequentially. However, the drug releases profiles in vitro and in vivo efficacy were not evaluated. Antitumor agent 10 hydroxycamptothecin (HCPT) was conveyed by a covalently modified MWCNT [128]. Diaminotriethylene glycol was used as a spacer linking the carboxylic groups on the oxidized nanotube to drug molecules via amidation reaction. The MWCNT formulated HCPT showed an improved antitumor activity both in vitro and in vivo compared to the clinical HCPT formulation and the MWCNT–HCPT conjugates have a longer blood circulation and a higher drug accumulation at the tumor site [137]. In a different approach, chemically functionalized SWCNTs have shown promise in tumor targeted accumulation in mice and exhibit biocompatibility, excretion and little toxicity. On this base, SWCNTs were first functionalized with branched poly (ethylene glycol) chains; afterwards, PTX was linked to the branched of PEG via amide bonds [119]. The extent research reveals that conjugated paclitaxel (PTX), a widely used cancer chemotherapy drug to branched polyethylene-glycol chains on SWCNTs via a cleavable ester bond to obtain a water soluble SWCNT-paclitaxel conjugate (SWCNT-PTX). SWCNT-PTX affords higher efficacy in stifling tumor growth than clinical paclitaxel in a murine 4T1 breast-cancer model, owing to prolonged blood circulation and 10-fold higher tumor PTX uptake by SWCNT delivery likely through enhanced permeability and retention effect. The SWCNT–PTX complexes were shown to stay in the blood for longer times (81.4±7.4 min) than drug (18.8±1.5 min) and PTX coated with PEG (22.8±1.0 min). This prolonged circulation time together with EPR effects allowed for much higher accumulation of the drug at tumor sites (10-fold higher than PTX and 6-fold higher than PEG–PTX after 2 h of injection and 6- and 4-fold higher, respectively, after 24 h of injection) [109]. In conclusion, the high delivery efficacy of PTX by SWCNT subsequently resulted in tumor inhibition of SWCNT–PTX (5 mg/kg PTX) for the 4T1 tumor model known to be resistant to PTX treatments [138-143]. It has been shown that nanotube drug delivery is promising for high treatment efficacy and minimum side effects for future cancer therapy with low drug doses. In another study, SWCNTs have been a pre-functionalized covalently with PTX-succinic anhydride-modified, and folic acid, as a targeting agent for many tumors toward cancer cells in order to open up new opportunities in chemical, biological, and medical applications of novel nano-materials (Figure 3) [50]. In this approach both the FA and PTX covalently functionalized upon amide- SWCNTs were well-solubilized and stable in water, PBS, and cell medium containing 10% fetal calf serum and full serum and biocompatibility and high targeting ability. Atomic force microscopy was used to measure the diameter and length of the functionalized SWCNTs after deposition on a Si substrate. Moreover, in order to insert the covalently conjugated onto to modified SWCNTs prepared by 1-(3-(dimethylamino) propyl)-3-ethylcarbodiimide hydrochloride (EDC) and N-hydroxysuccinimide (NHS) [144,145]. It is suggested that the drug was successfully loaded onto the SWCNTs, which may be mainly broken up by covalent stacking and hydrophobic interactions. The concentration of PTX loaded onto SWCNTs was measured by the absorbance peak at 240 nm with a molar extinction coefficient of 31.7 ×105 M.cm -1. Loaded onto the nanotubes the quantity of PTX was measured by UV-Vis-NIR spectra, for the same batches of samples [50]. Since the application of PTX in clinical therapy is limited by poor aqueous solubility, inefficient distribution, and the lack of selectivity, modification of PTX with succinic acid (the cleavable linker) CNTs exhibited good aqueous solubility, biocompatibility [144]. According to the latest results polymers change the physicochemical property of CNTs in biological medium and ability of polymers to develop the efficacy of CNTs as drug delivery systems. It is the consequence of secondary interactions among these two portions. The superior mechanic strength and chemical inert also makes it ideal for blood compatible applications, Moreover the functionalized CNTs have been extensively explored for in vivo targeted drug, which will potentially improve the efficiency of many cancer treatments (Figure 4) [50].
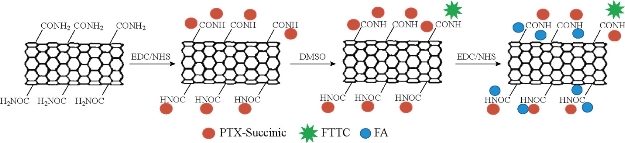
Fig.3. The preparation of SWCNT with targeting properties involves several steps: coating SCNTs with paclitaxel, attaching Fluorescein isothiocyanate probe to the SWCNT-PTX, and finally targeting molecules (FA) attachment [50].
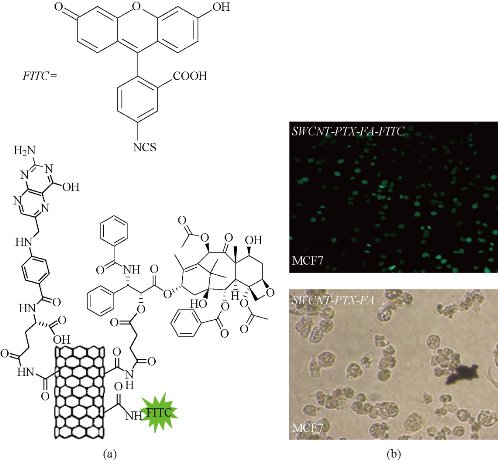
Fig.4. (a) Chemical formula of the SWCNT/PTX–FA/FITC (SWCNTs functionalized with paclitaxel and folic acid at on the sidewall by covalent bond); (b) confocal fluorescence images of MCF-7 breast cancer cells treated with SWCNT/PTX/FA [50].
Carbon Nanotubes - Magnetic Nanoparticles Assemblies
Over the past few years, researchers have developed various types of nanoparticles for treatment of cancer [146-151]. Magnetic nanoparticles have received pervasive attention in medicine communities owing to their potentials in medical applications [146-151]. These applications associate closely with the unique magnetic properties of MNPs with comparable size to biologically important objects. Magnetic CNTs have also illustrated favorable outcomes as a MRI contrast agent with high nuclear magnetic resonance relaxivities, little cytotoxicity, catalysis [152], magnetic hyperthermia [153] and high cell-labeling efficiency [154]. As mentioned earlier, CNTs possess also a hollow cavity that can be filled with a variety of metals such as Au, Ag, Cu, Sn, Fe, Co, and Ni and employed as nano-antennas or microscopic probes [154-156]. These main reasons evolve in many studying in order to functionalize (or coating) of CNTs such as magnetic or super paramagnetic nanoparticles or in filling their cavity with magnetic molecules in order to obtain versatile systems being able to be employed more efficiently in biomedical or bio-imaging applications. In fact, the biological molecular could be tagged on the MNPs, which are monitored and manipulated in the presence of external magnetic fields [157-160]. The chemical combination of magnetic nanoparticles or nano-crystals and CNTs in order to obtain nano-hybrid structures, follows various strategies including, encapsulation of magnetic molecules inside the carbon nanotubes or grafting/decorating CNTs on their surface by bio-conjugation chemistry or electrochemical deposition. The strategies to attach some ready synthesized nano-crystals have been achieved using covalent bonds [159], electrostatic interactions [158], π-π stacking [159], and hydrophobic interactions [160]. Many other strategies have been devised in the last few years and we will discuss separately their applications, according to their various application.
Covalent method
One of the covalent attachment strategies [161] has received much interest owing to the possibility of grafting gold nano-rods and multi walled carbon nanotubes and developed a simple and effective strategy to prepare NIR absorption enhancer MWCNTs through covalent interaction of carboxyl groups on the MWCNTs with silica-coated gold nano-rods (sGNRs). Then, modified Gold nanorods peptides with the amino silane agent were conjugated with the sGNR-MWCNT hybrid structure in a struggle to eliminate their cytotoxicity and improving their biocompatibility, as a result of which RGD-conjugated sGNR-MWNT (RGD-GNR-MWNT) nano-probes were used for photoacoustic imaging of in vivo gastric MGC803 and GES-1 cells. Gold nano-rods, due to their small size, strong light-enhanced absorption in the NIR, and plasmon resonance-enhanced properties have become attractive noble nano-materials for their potential in applications including, photothermal therapy [162], bio-sensing [163], PA imaging [164], and gene delivery [165] for cancer treatment. According to the cell viability data RGD-GNR-MWNT nanoprobes revealed good biocompatibility to MGC803 and GES-1 cells within the dose of 800 µg/mL in the medium. These results illustrated that RGD-conjugated sGNRs/MWNTs may be a good contrast agent for photoacoustic imaging of in vivo gastric cancer cells, and gold nano-rods can enhance the photoacoustic (PA) signal of MWNTs. Golden single-walled carbon nanotubes have been used for photoacoustic imaging of in vivo tumors [166,167]. Additionally, regarding that RGD-conjugated sGNR/MWNT probes exhibited good water solubility and low cellular toxicity, it could target in vivo gastric cancer cells, and obtained strong photoacoustic imaging in the nude model. In a recent work, the application in cancer targeted imaging and magnetically guided drug delivery was reported by Yang et al. [75], where a novel magnetic lymphatic-targeting drug-delivery system, based on functionalized carbon nanotubes, was presented in order to improve cancertherapy with lymph node involvement. Multiwalled carbon nanotubes have been covalently functionalized with magnetic poly-acrylic acid (PAA) in order to obtain MWCNTs-GEM and deliver the targeted cell of this novel lymphatic (Figure 5(a)). Gemcitabinis (GEM) a nucleoside analogue and a S-phase specific cytotoxic agent shown activity in many solid tumors, such as pancreatic cancer, non-small cell lung cancer, head and neck squamous cell cancer, germ cell tumors, and tumors of the bladder, breast, ovary, cervix and biliary tract, as well as some hematologic malignancies [76]. It is suggested that targeting drug delivery system using functionalized multiwalled carbon nanotubes can be effective for cancer treatment. The mMWCNTs/GEM was subcutaneously injected into a mouse that had cancer lymph node metastasis via the left rear footpad, and were taken up into lymphatic vessels and retained in the targeted lymph node under the magnetic field (Figure 5(b)). The in vitro cell cytotoxicity after various treatments was assessed at given time functionalized PAA-MWCNTs designed with magnetite nanoparticles (Fe3O4) can be efficiently taken up by lymphatic vessels and delivered to regional lymph nodes in vivo with little toxicities. In addition, this magnetic lymphatic targeting system [77] can deliver gemcitabine into the lymph nodes in vivo with high efficiency under the guidance of magnetic field [168].
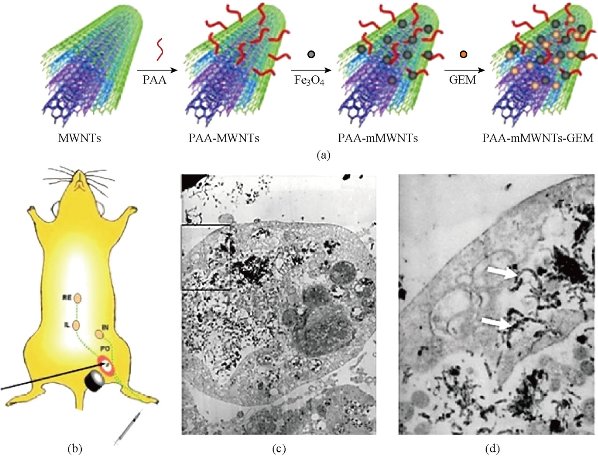
Fig.5. The preparation of the Magnetic lymphatic drug delivery system, (a) magnetic lymphatic targeted chemotherapy in mice and illustration of chemical reactions used to attach gemcitabine onto mMWCNTs; (b) magnetic lymphatic targeted chemotherapy in mice. MMWCNTs/GEM was subcutaneously injected into a mouse that had cancer lymph node metastasis and TEM image (c) and (d) of mMWCNTs-treated human pancreatic cancer cell line. The white arrows point to mMWCNTs inside the cytoplasm [75].
Non-covalent method
Recently, groundbreaking efforts have been made for fabricating magnetic carbon nanostructures by conjugating MNPs with carbon nanotubes (CNTs) [169]. However, the attachment of iron oxide nanoparticles and QDs on the active surface of CNTs considerably plummeted the CNTs capacity for drug storage and further functionalization [170,171]. Additionally, direct contact of Quantum dots with iron oxide nanoparticles on CNTs surface causes fluorescence quenching and diminishes the magnetic-fluorescent dual functionality. An effective method to incorporate CdTe quantum dots with Fe2O3 -filled carbon nanotubes (CNTs) with different capping agents onto the surface of functionalized carbon nanotube has been developed for the first time [64]. In this work, Fe2O3 is filled the hallow cavity of CNTs facilitating magnetically guided delivery and improves the synergetic targeting efficiency [64]. MNPs were encapsulated into the interior cavity of CNTs with the aim of diminishing the magnetic-fluorescent dual functionality and minimize the potential toxic side-effects by exposure of free magnetic nanoparticle to biological environment [64,65]. Moreover, the biocompatible and chemically inactive hybrid SiO4 shells provide a cover layer to mitigate the toxicity of Quantum dots and to protect its fluorescence from being quenched by CNTs; afterwards, poly (sodium 4-styrenesulfonate) (PSS) was absorbed onto the surface of modified magnetic CNTs via π-π interactions, afterwards non-covalent interactions between poly allylamine (PHA)-Hybrid SiO2-coated QDs (HQDs) nanoparticles and PSS-coated Fe2O3/CNT have resulted in preparation of Fe3O4/ CNT-HQDs. The CdTe nanoparticles were coated with a thin SiO2 layer containing Cd2+ and thioglycolic acid (TGA). Fe3O4/CNT-HQDs-Trf forms through the interaction between formed amine-reactive Trf-NHS ester and Fe2O3/CNT-HQDs by amide bond. Eventually, doxorubicin loaded onto Fe3O4/CNT-HQDs-Trf via π-π interactions. The efficacy of drug delivery systems for killing the cancer cells and targeting the drugs towards tumors has been investigated. Interestingly, in this work, doxorubicin released from Fe3O4/CNT-HQDs-Trf is highly pH-dependent, and a very limited amount of DOX is released at pH 8.5 and 7.4. It is of vital importance that the selective delivery of CNTs to tumor cells- through the interaction between transferrin and receptors of the Hela cells- minimizing their side effects can be achieved by simultaneously anchoring iron oxide nanoparticles and Fe3O4/CNTHQDs-transferrin followed by using an external magnetic field. It is noticeable that the uptake of Fe3O4/CNT-HQDs-Trf is further remarkably improved, for in the presence of an external magnetic field [172,173].
Toxicity of Carbon Nanotubes
It is of vital importance to determine carbon nanotubes’ pharmacological and toxicological profiles in order to successfully apply a new generation of nano-materials as nano-medicine [173]. Successful interpretation of these results is difficult because the CNTs can significantly disrupt the emission of the certain particles [174-178]. Although the molecular and cellular mechanisms for the cytotoxicity and the entering of CNTs into cells have not been completely understood, functionalized carbon nanotubes are successfully able to cross cell membranes via an endocytosis pathway or endocytosis independent mechanism. Toxicity and pathology engendered by CNTs on an organismal in both in vivo and in vitro studies have been attributed to various factors in order to determine the nature of nano-toxicity in living organisms and cells including number of walls, length and aspect ratio, surface area, degree of aggregation, rigidity, extent of oxidation or functionalization, purity, hydrophobicity, surface topology, method of administration, dispersibility, type and degree of functionalization, and method of manufacture (which can leave catalyst residues and produce impurities) [28]. Additionally, toxicity of CNTs is also dependent on their concentration, dose, duration and method that cells or organisms are exposed to and even the utilized dispersant to solubilize the nanotubes. The length and shape of CNTs influence how well they cross the membrane of macrophages and determine the resulting immunologic response [64,172]. For example, following synthesis, residual metal (Co, Fe, Ni, and Mo) is normally encapsulated in a layer of carbon, either amorphous soot or layers of graphite [179]. It is strongly suggested that one way of doing so is toss ultra-sonication owing to the fact that ultra-sonication promotes the release of a metallic impurities into solution [180]. It has been discovered that metal impurities is one of the main factors that determines CNTs toxicity, resulting in cell death through various mechanisms such as mitochondrial destruction and oxidative stress [181]. To that end, scientists have examined CNT toxicity in several cell lines [182,183], as well as using in vitro studies to divulge the effect of various factors on CNT toxicity, in vivo studies have also played a major role for investigating the CNT toxicity. Regardless of the drawbacks of in vitro studies, they have been invaluable in identifying important side effects of CNTs. In vivo studies are favored over in vitro studies, as long as they allow nanotoxicity to be measured on an entire organism, rather than in cell culture. Compared to pristine CNTs, well-functionalized CNTs by biocompatible coatings such as PEGylation exhibit remarkable reduction of toxicity in vivo after being intravenously injected into animals [82,184-188]. Nonetheless, the extent research reveals that as well as others, specific types of functionalization CNTs can greatly minimize CNT toxicity and represent promising progress towards their pharmacological applications.
Conclusions
In summary, this study discusses some chemical methods to modify CNTs for the drug delivery. An ideal CNT is expected to enter and destroy cancer cells while minimizing the accumulation and side effects to normal organs. CNTs appear to accumulate in cancerous tumor tissue much more than in normal tissues, partly due to the EPR effect. The reported data indicate a potential of modified CNTs in different biomedical applications, particularly as a valuable tool in anticancer drug delivery due to their good aqueous solubility and biocompatibility. It is obvious that polymers change the conformation and shape of CNTs from strand-type toward closed conformations. Finally, investigations on all possible ways for interactions of CNTs and polymers lead to better knowledge in the design of novel anticancer drug delivery systems.
Competing Interests
The authors declare that they have no competing interests.
References
Copyright© 2016 Sara Tavakolifard, Esmaeil Biazar. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.